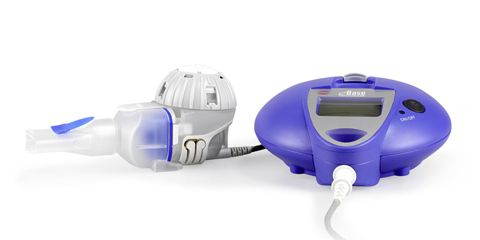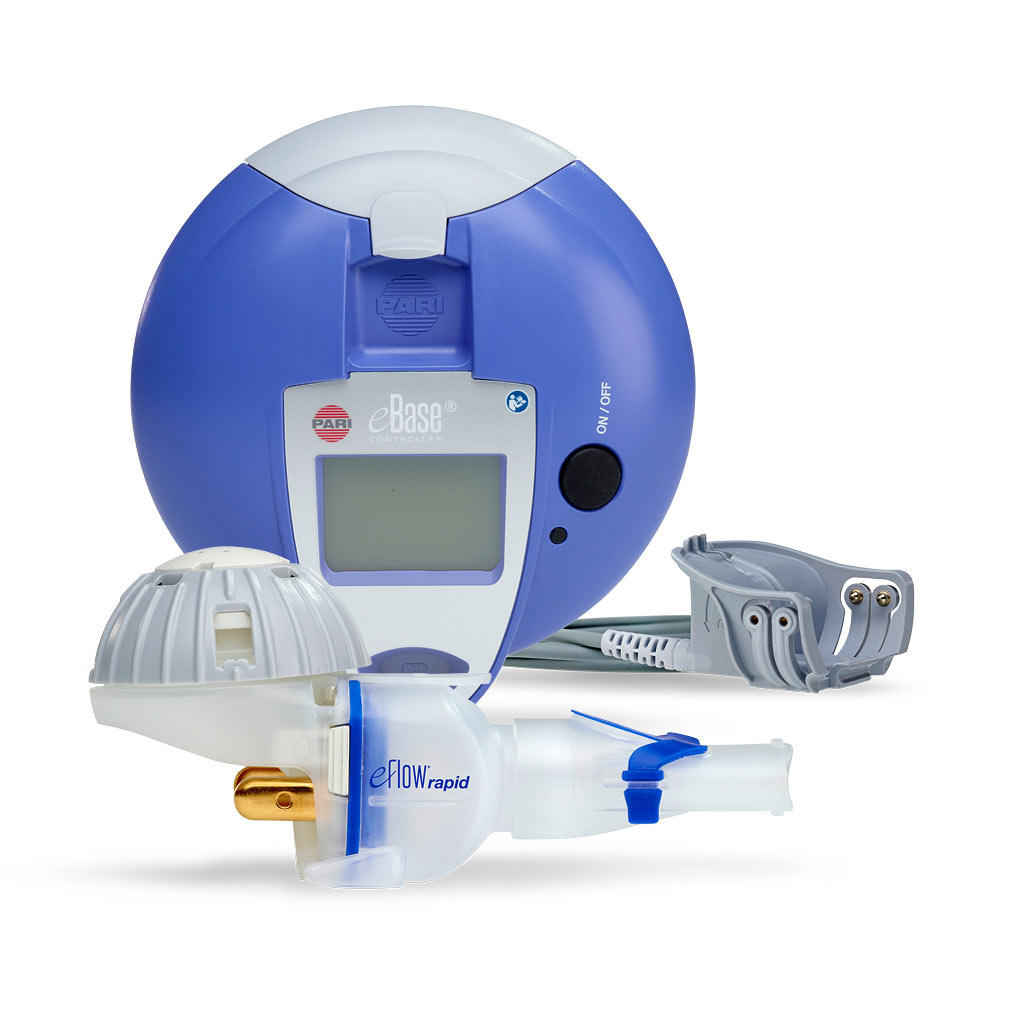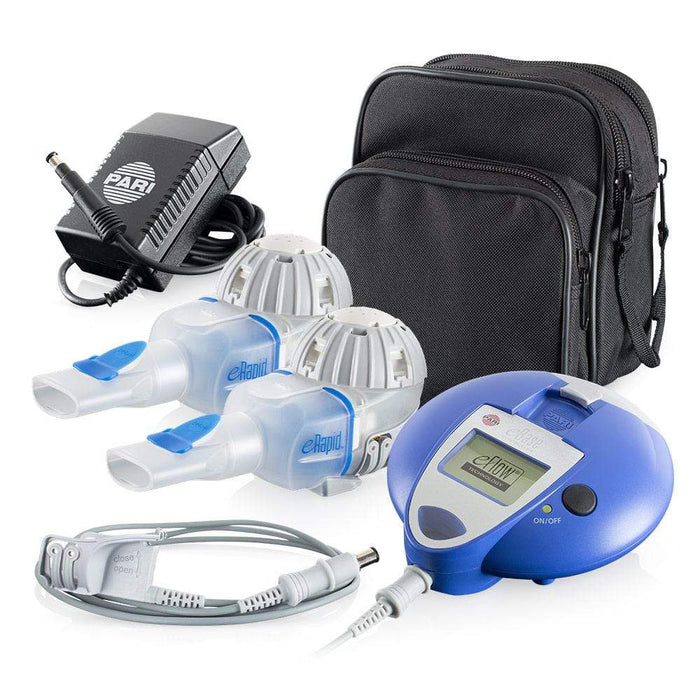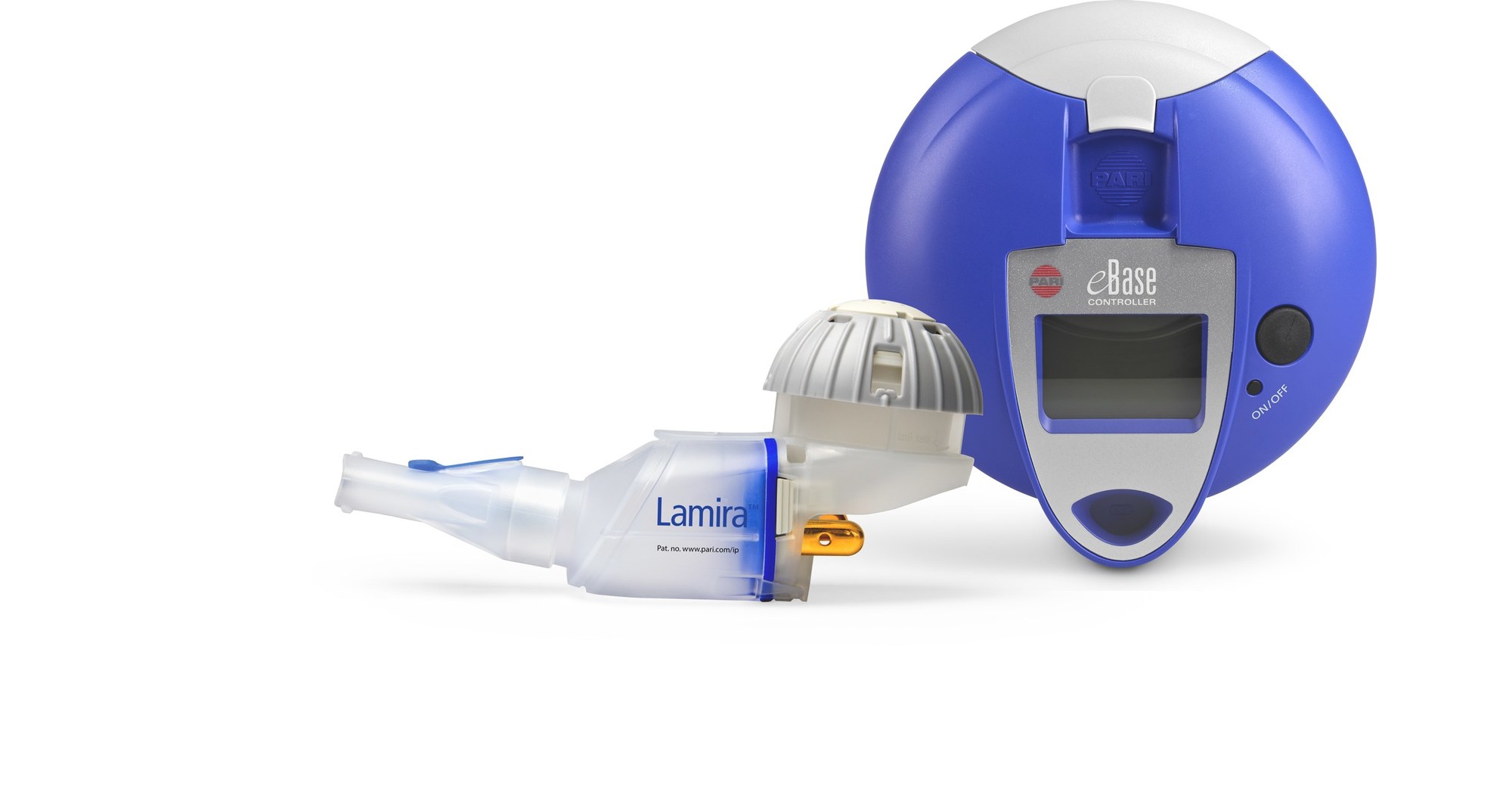
PARI Pharma's eFlow® Technology device, LAMIRA™, approved as the only nebulizer system to deliver Insmed's ARIKAYCE® (amikacin liposome inhalation suspension)

The Pari eFlow (called APIXNEB when used with the AKITA) open-mesh,... | Download Scientific Diagram