If the relative atomic weight scale is changed from 1/12 to 1/6th the mass of carbon-12, the mass of 1 mol of a substance wil Single Correct MCO 4 marks

5 The Structure of Atoms. 2 Chapter Outline Subatomic Particles 1.Fundamental Particles 2.The Discovery of Electrons 3.Canal Rays and Protons 4.Rutherford. - ppt download
68. Atomic weight of calcium is 40amu on C 12 scale.If 1/10 of an atom of C 12 is taken as standard then relative atomic weight of calcium will be ?

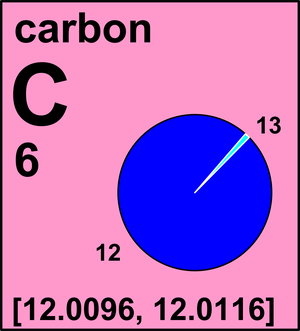


:max_bytes(150000):strip_icc()/atomic-weight-and-atomic-mass-difference-4046144_FINAL_STILL-5940e35000b145ba83fb8e3e40792ba9.png)



:max_bytes(150000):strip_icc()/boron-illustration-545864379-5838819f5f9b58d5b1c57b5f.jpg)

![The modern atomic weight scale is based on [MP PMT 2002] The modern atomic weight scale is based on [MP PMT 2002]](https://blogger.googleusercontent.com/img/b/R29vZ2xl/AVvXsEhe1xivZzme3VvaXcGUWU7j8rzWTfKM0SMHKY82JBF73rIq1OUsBzJmBPfRNTHDNofx-GCYVo5fEoy--WXE4p7mHMQBjkW8z6t3zAb08-qYDDGNp7Z2kB5lyumCF_XhTbeHyrmv6JDlKD5Wrq_PyVTAJQ9nU-uwDw0zsLC_fcJoWc3fOYBtFj93Omfw/w1600/The%20modern%20atomic%20weight%20scale%20is%20based%20on%20%5BMP%20PMT%202002%5D.jpg)